Prologue on seeds.
In the introduction page, the topic of the site - cumulative-dissipative processes - was briefly presented.
In this prologue on seeds, some points will be presented that I want to highlight, among those that will then be treated in detail in three specific itineraries.
1.0.#1 - A sowing procedure to increase yields.
1.0.#2 - How to improve seed germination.
1.0.#3 - How the cumulative-dissipative processes are activated.
1.0.#4 - Short episodes of interaction.
1.0.#5 - Sowing calendars.
1.0.#6 - Efficiency of the cumulative-dissipative cycle in seeds.
1.0.#7 - Seven lean years.
1.0.#8 - In the open field; in a greenhouse.
---- 1.0.#1 ----
A sowing procedure to increase yields.
I have developed a planting method, aimed at increasing yields, and which I intend to make public.
---- 1.0.#2 ----
How to improve seed germination.
From what I have been able to understand so far, seeds manage their germination capacity, in order to maintain it for a long time, thanks to cumulative-dissipative processes.
Usually, these processes take place with a reduced efficiency, because they are left to chance. A procedure, which seems paradoxical, allows to make them more efficient.
It is as if a little increase in entropy, during the cumulative phase, favors a greater reduction, during the subsequent dissipative phase.
In fact, during the cumulative phase a sort of decomposition takes place, a necessary step to allow recomposition in the subsequent dissipative phase, and to return to a previous low level of entropy.
In a certain sense, it's like erasing the effects of the passage of time and starting over again.
Indeed, it is the “force d” (force due to the angular motion with respect to other matter) that changes the logic to be used, as we will see better in the three itineraries of discovery on the seeds.
---- 1.0.#3 ----
How the cumulative-dissipative processes are activated.
Cumulative-dissipative processes take place in seeds in such peculiar ways, that it is as if they had their own signature.
As I saw in “experiment A” (Sunflower seeds in motion with respect to the surrounding matter) and in “experiment E” (Seeds stationary with respect to the surrounding matter, but in motion with respect to the Moon), these processes are activated:
(1) by angular movement with respect to other matter;
(2) and by exchanges of heat, which is first lent to the seed, when said movement increases (cumulative phase), and then returned by the seed, when said movement decreases (dissipative phase).
I deduced this from the fact that the phenomena are more evident when the angular motion with respect to other matter persists for a long time at specific values of angular velocity.
To confirm what is written here, further research is necessary, with the use of adequate instrumentation, which I lack.
---- 1.0.#4 ----
Short episodes of interaction.
All this appears to occur only during brief episodes of interaction, when the motion is at one of the critical angular velocities.
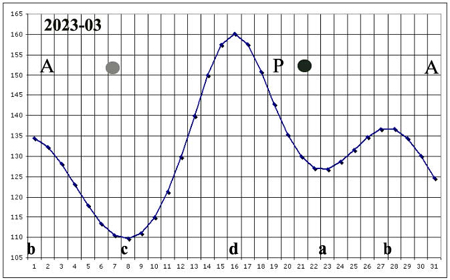
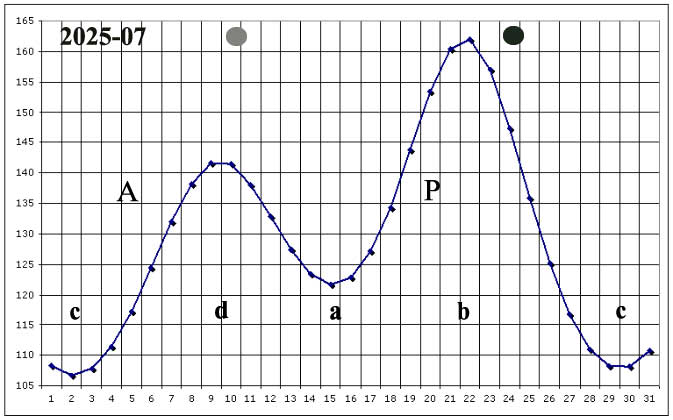
I have been able to witness some of these episodes when the variation of the Moon's angular velocity with respect to the Earth is small, which generally happens near points a, b, c, d of the calendar of these velocities.
I deduced this from the fact that the phenomena are more evident when the angular motion with respect to other matter persists for a long time at one of the specific values of angular velocity, which appears to be somehow critical.
To confirm what is written here, further research is necessary, with the use of adequate instrumentation, which I lack.
---- 1.0.#5 ----
Sowing calendars.
In presenting the sowing procedure, intended to improve the germination capacity of seeds, I use calendars, where it appears when cumulative and dissipative processes can be underway in the seeds, when they are stationary with respect to the Earth.
In this site, in order to make it easier to read, in my opinion, the sowing calendars, given for each month of the normal calendar, indicate the hourly angular velocity of the Moon, in its rotation around the Earth, defined in 86,400 deltins, and performed in a sidereal month, and not the angular velocity of the seeds with respect to the Moon.
Consequently, the cumulative phases take place when said movement is indicated as decreasing (periods b-c; d-a), while the dissipative phases take place when said movement is indicated as increasing (a-b; c-d).
---- 1.0.#6 ----
Efficiency of the cumulative-dissipative cycle in seeds.
The cumulative-dissipative cycle, thanks to which seeds maintain their ability to germinate over time, varies in efficiency for several reasons.
The most important is the variation in the Moon's declination relative to the equator. The less it varies, the better.
Over the course of 18.6 years, this declination can range from just over 14 degrees to 28.5 degrees.
Over the course of these 18.6 years, the recommended sowing procedure increases crop yields compared to what would normally be achieved.
In a high-yield year, this increase is 30 percent; in a low-yield year, the increase is 50 percent.
Examples.
1.1.5 - Harvest result in a year of lean times (2005).
1.1.6 - Harvest result in a year of abundance (2015).
The variability in the efficiency of cumulative-dissipative processes can be explained roughly by the fact: (1) that they only take place at critical angular velocities with respect to other matter; (2) the greater the declination with respect to the equator, the shorter the times during which the seeds remain at a useful critical angular velocity. Of course, it is also related to the quantity of heat exchanges, in a cumulative sense if there is an increase in angular velocity, in a dissipative sense if there is a decrease in said velocity.
---- 1.0.#7 ----
Seven lean years.
When the variation of the declination of the Moon with respect to the equator exceeds 26 degrees, during seven years within the cycle, the efficiency of the cumulative-dissipative processes is reduced.
In fact, due to the fact that the processes occur only at critical angular velocities, the more the declination of the Moon varies per unit of time, with respect to the Earth's equator, the shorter the episodes during which the cumulative-dissipative processes can take place, the lower their efficiency.
When, in the cumulative phase, said episodes are short, and at the same time the temperature is low, the efficiency of the cycle is compromised, as some natural food crises, i.e. those not due to wars, testify.
A combination that has caused the most serious famines throughout history.
---- 1.0.#8 ----
In the open field; in a greenhouse.
All the experiments on seeds, published on this site, were carried out in the open field, not in a greenhouse.
However, in order to respect the cycle times, the procedure is best performed where the timing of the water supply can be managed, as can take place in a greenhouse, rather than being dependent on the vagaries of the weather.
Soil depletion.
In order to avoid soil depletion, the procedure also requires a suitable rotation of crops, alternating improving species, preparatory species and depleting species.
This would also allow for lower costs in terms of plant protection products and fertilizers.
In this regard, the use of fossil-based fertilizers should be avoided. First, because they are harmful to quality and the environment (especially in the long term, due to their greenhouse effect). Second, because they only provide a portion of the necessary elements. It is those not supplied that limit the effectiveness of the fertilizers provided.
continued index seeds